Bacterial Cell Walls
There Are Two Main Types of Bacterial Cell Walls, The cell wall is the layer that lies just outside the plasma membrane.
It is one of the most important structures for several reasons:
- It helps maintain cell shape and protect the cell from osmotic lysis; it can protect the cell from toxic substances; and in pathogens, it can contribute to pathogenicity.
- Cell walls are so important that most bacteria have them.
- Those that lack them have other features that fulfill cell wall function.
- Bacterial cell wall synthesis is an important target for many antibiotics.
Overview of Bacterial Cell Wall Structure
After Christian Gram developed the Gram stain in 1884, it soon became evident that most bacteria could be divided into two major groups based on their response to the Gram-staining procedure. Gram positive bacteria stained purple, whereas Gram-negative bacteria were pink or red.
The true structural difference between these two groups did not become clear until the advent of the transmission electron microscope. Here we describe the long-held models of Gram-positive and Gram-negative cell walls developed from these studies.
More recent studies of diverse groups of bacteria have shown that these models do not hold true for all bacteria Because of ongoing discussions related to these new studies, we will refer to bacteria that fit the models as being typical Gram-positive or typical Gram negative bacteria.
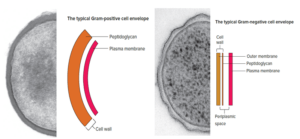
The cell walls of Bacillus subtilis and many other typical Gram-positive bacteria consist of a single, 20- to 80-nm-thick homogeneous layer of peptidoglycan (murein) lying outside the plasma membrane. In contrast, the cell walls of E. coli and many other typical Gram-negative bacteria have two distinct layers: a 2 to 7nm thick peptidoglycan layer covered by a 7 to 8nm thick outer membrane.
One important feature seen in typical Gram-negative bacteria is a space between the plasma membrane and the outer membrane. It also is sometimes observed between the plasma membrane and cell wall in typical Gram-positive bacteria. This space is called the periplasmic space. The substance that occupies the periplasmic space is the periplasm.
Peptidoglycan Structure
The feature common to nearly all bacterial cell walls is the presence of peptidoglycan, which forms an enormous mesh like structure often referred to as the peptidoglycan sacculus.
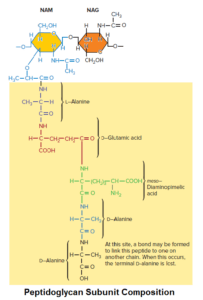
Peptidoglycan is composed of many identical subunits. Each subunit within the sacculus contains two sugar derivatives, N-acetylglucosamine (NAG) and N-acetylmuramic acid (NAM), and several different amino acids.
The amino acids form a short peptide, sometimes called the stem peptide, consisting of four alternating D- and L-amino acids; the peptide is connected to the carboxyl group of NAM.
Three of the amino acids are not found in proteins:
- D-glutamic acid
- D-alanine
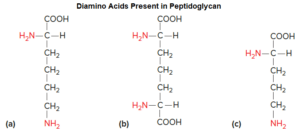
- Meso-diaminopimelic acid.
The presence of D-amino acids in the stem peptide protects against degradation by most peptidases, which recognize only the L-isomers of amino acid residues. The peptidoglycan subunit of many bacteria is shown in figure.
The peptidoglycan sacculus is formed by linking the sugars of the peptidoglycan subunits together to form a strand; the strands are then cross-linked to each other by covalent bonds formed between the stem peptides extending from each strand.
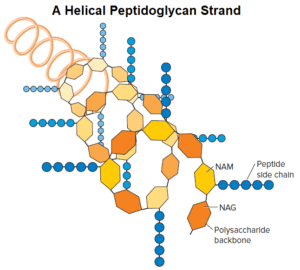
As seen in figure, the backbone of each strand is composed of alternating NAG and NAM residues. The strand is helical, and the stem peptides extend out from the backbone in different directions. There are two types of cross-links: direct and indirect via a peptide inter bridge.
A direct cross-link is characterized by connecting the carboxyl group of an amino acid in one stem peptide to the amino group of an amino acid in another stem peptide.
For instance, many bacteria cross-link the strands by connecting the carboxyl group of the D-alanine at position 4 of the stem peptide directly to the amino group of diaminopimelic acid (position 3) of the other peptidoglycan strand’s stem peptide (the position 5 d-alanine is removed as the cross-link is formed).
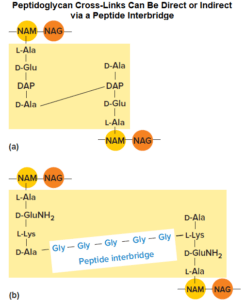
Bacteria that have indirect linkage use a peptide inter bridge (also called an interpeptide bridge), a short chain of amino acids that links the stem peptide of one peptidoglycan strand to that of another. Cross-linking results in one dense, interconnected network of peptidoglycan strands.
The peptidoglycan sacculus is strong but elastic. It is able to stretch and contract in response to osmotic pressure. This is due to the rigidity of the backbone coupled with the flexibility of the cross-links. Peptidoglycan sacculi are also rather porous, allowing globular proteins having a molecular weight as large as 50,000 to pass through, depending on whether the sacculus is relaxed or stretched; thus only extremely large proteins are unable to pass through peptidoglycan.
Variants of peptidoglycan are found, particularly among typical Gram-positive bacteria. For example, some substitute the diamino acid lysine for meso-diaminopimelic acid and cross-link chains via interpeptide bridges. These interpeptide bridges can vary considerably.
Peptidoglycan can also vary in terms of the length of the peptidoglycan strands and the amount of cross-linking. Bacteria that stain Gram positive tend to have much more cross-linking, whereas those that stain Gram negative have considerably less.
References and Sources:
- https://quizlet.com/513775513/chp-2-fig-notes-micro-flash-cards/
- https://www.mheducation.com/highered/ideas/i/1169475-flipbook2019-willey-prescottsmicrobiology-11e/38
- https://quizlet.com/151347682/microbiology-ch-3-flash-cards/
- https://en.wikipedia.org/wiki/Bacterial_cell_structure
- https://quizlet.com/213308873/introductory-microbiology-exam-cell-wall-and-gram-stainflash-cards/
- https://academic.oup.com/femsre/article/32/2/149/2683904
- https://link.springer.com/article/10.1007/s00726-017-2459-5
Also Read:
- Agglutination reaction: Definition, Uses and Application
- Mycobacterium tuberculosis-Epidemiology, Pathogenesis, and Treatment
- Cell Cycle: Introduction, Phase, Mechanism and Significance
- Different Staining Methods used in Microbiology
- Plasma Membrane- Structure, Properties, Lipid Rafts and Glycolipids
- Difference between Prokaryotes and Eukaryotes
- Comparison Between the Domains Bacteria, Archaea, and Eukarya
