Introduction
- Gel electrophorisis is simple, rapid and sensitive analytical technique for the separation of charged particle.
- The gels, however, are porous and the size of the pores relative to that of the molecule determines whether the molecule will enter the pore and be retarded or will bypass it. The separation thus not only depends on the charge on the molecule but also on its size. Needless to say, that resolution of a sample is sharper and better in a gel than in any other type of medium.
- Agrose gel is used as a supporting media for the separation of DNA, RNA or protein under the influence of electric charge.
- Most of the biomolecules has a net charge at any pH other than at their isoelectric point.
- There is difference in the electrophoretic mobility of these charged molecules due to their difference in size, shape, and charge.
There are basically two types of materials are used to make gels:
- Agrose
- Polyacrylamide
Agarose
- Agarose is natural colloid which is isolated from the seaweed.
- It is linear polysaccharide.
- It is made up of repeating units of agarobiose, comprises alternating units of 3,6-anhydrolactose and galactose.
- This gel has generally larger pore size, which makes them suitable to separate larger molecules having molecular mass more than 200 kDa.
- It is most commonly used for the electrophoresis of both protein and nucleic acids.
- Agarose is used in concentration between 1% and 3%.

Polyacrylamide gel
- Polyacrylamide gel is consisting of chains of acrylamide monomers crosslinked with N, N’-methylenebisacrylamide units, which is commonly termed as bisacrylamide.
- In this gel, pore size and resolving power is totally depends upon the concentration of acrylamide and bisacrylamide.
- The concentration of the gel normally varies from 5% to 25%.
- This gel is used in electrophoresis for the separation of proteins ranging from molecular weight <5000 to >200,000, and polynucleotides ranges from <5 to ~ 3000 base pairs in size.
Apparatus of gel electrophoresis
- Vertical gel apparatus: It is commonly used IN sds PAGE for the separation of proteins.
- Horizontal gel apparatus: It is used for immune electrophoresis, iso-electric focusing and electrophoresis of DNA and RNA in the agarose gel.
Type of gel electrophoresis
- Agarose gel electrophoresis
- SDS-PAGE
- Pulse field gel electrophoresis (PFGE)
- 2D gel electrophoresis
Agarose gel electrophoresis
- Agarose gel the supporting media in the electrophoresis.
- For the electrophoresis of DNA, RNA and Protein agrose gel is used.
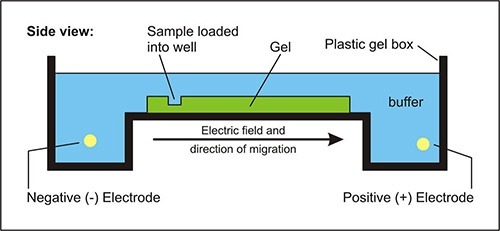
Principle of gel electrophoresis
In the agrose gel electrophoresis the potential difference is applied across the electrodes in a horizontal electrophoretic tank containing agarose gel and biomolecules (such as nucleic acid or proteins) is loaded, then molecules migrated to their respective electrodes. The rate of migration of charged particles depends on the size, shape, molecular mass etc.
In this process, larger molecules have difficulty in moving through the pore size of the supporting media, whereas the smaller molecules has more mobility through it. The bands of protein or nucleic acid is visualized by using intercalating dye, i.e., ethidium bromide (Etbr), they are visualized by fluorescence when illuminated with ultraviolet lights.
Requirement/ instrumentation:
- An electrophoretic unit,
- A power supply,
- Gel casting trays
- Combs
- Agarose gel or media
Electrophoresis buffer
Composition and ionic strength of electrophoresis buffer is most important factor for the separation of nucleic acids (DNA or RNA).
Most routinely used buffers are:
- TAE- (Tris-acetate-EDTA), it has lower buffering capacity and generally used to separate larger nucleic acid fragments (>12kb).
- TBE- (Tris-borate-EDTA), it has high buffering capacity and higher ionic strength and generally used for the separation of low molecular weight compound (<1kb).
- Loading buffer: Nucleic acid is before loading on to a gel is first mixed with the gel loading buffer, which usually consists of:-
- Salts: It creates environment with favorable ionic strength and pH of the sample, e.g., Tris-HCl.
- Metal chelator: It prevents nucleases to degrade the nucleic acid such as EDTA.
- Loading dyes: It provides color for tracking and easy monitoring of sample. Such as, bromophenol blue, xylene cyanol.
- Transilluminator: (An ultraviolet light box), which is used to visualize bands in gels.
Applications
- In molecular genetic diagnosis or genetic fingerprinting for analysis of PCR products.
- For the estimation of size of DNA molecule.
- In the separation of restricted DNA and RNA.
- In addition to providing an excellent medium for fragment size analyses, agarose gels allow purification of DNA fragments.
SDS-PAGE Electrophoresis
- Sodium dodecyl sulfate (SDS) polyacrylamide gel electrophoresis is routinely used for the separation of proteins on the basis of their mass.
- It involves the use of vertical gel apparatus to separate proteins.
Principle of SDS-PAGE
This technique uses anionic detergent sodium dodecyl sulfate (SDS) which disassociates proteins into their individual polypeptide subunits and gives a uniform negative charge along each denatured polypeptide. When these denatured polypeptides are loaded at the cathode end of an electric field, then we get clear bands of proteins arranged in decreasing order of their molecular mass from the cathode to anode.
Requirement/ instrumentation
- Gel or media acrylamide solutions.
- Buffer system the separation and migration patterns of proteins in gel electrophoresis are determined by the chemical composition and pH of the buffer system.
Three basic types of buffers are required:
- Gel casting buffer to cast gel
- Sample buffer to prepare the sample
- Running buffer to fill the electrode reservoirs
- lower reservoir amine buffer with HCl (running gel)
- Upper reservoir amine buffer with glycine (stack gel)
- Staining or tracking dye
- Protein samples
- Molecular weight markers
- An electrophoresis chamber and power supply
- Glass plates (a short and a top plate)
- Casting frame
- Casting stand
- Combs
Applications
- For measuring molecular weight.
- In Peptide mapping.
- For Estimation of protein size.
- For Determination of protein sub-units or aggregation structures.
- For Estimation of protein purity.
- In Protein quantitation.
- In monitoring protein integrity.
Pulse field gel electrophoresis (PFGE)
- Conventional agarose gel electrophoresis cannot separate linear double stranded DNA molecules that have radius of gyration, which is larger than the pore size of the gel.
- However, with certain changes in the orientation of electric field with respect to the gel, large DNA fragments can be resolved.
- It is used to separate large DNA molecule by applying gel matrix as electric field that periodically changes direction.
- This technique is invented by Schwartz and Cantor in 1984.
- DNA fragments up to 10 mb can be separated by this technique.

Principle of Pulse field gel electrophoresis
As opposed to the continuous unidirectional electric fields applied in conventional gel electrophoresis, pulsed-field gel electrophoresis uses pulsed, alternating, orthogonal electric fields. When such a field is applied to a gel, large DNA molecules become trapped into their reptation tubes every time the direction of the electric field is changed.
These molecules remain immobile till they reorient t themselves along the direction of the new electric field. It is here that different DNA molecules adapt a behavior consonant with their respective sizes; large DNA molecules take a longer time to reorient themselves and are consequently uninformed, more in the new electric field as compared to the smaller DNA molecules.
Thus, all those molecules of DNA whose reorientation times are less than the period of the electric pulse can be fractionated in a size dependent manner. Factors, which are of extreme importance for determining the limit of resolution of pulsed-field gel electrophoresis are given below:
- The absolute periods of the electric pulses.
- The angles at which the two electric fields are applied to the gel.
- The relative field strengths of the two electric fields and the degree of uniformity of the two electric fields.
- The ratio of the periods of the electric pulses employed to generate the two electric fields.
Instrumentation
- The original apparatus used pulsed electric fields or perpendicular orientations and linear electrodes.
Applications
- It is used for the separation of DNA more than 10 mb.
- It generated stable and reproducible DNA restriction patterns.
- This technique applied to the sub-typing of many pathogenic bacteria and has high concordance with epidemiological relatedness.
- This technique can be applied as a universal generic method for sub-typing of bacteria.
2D gel electrophoresis
- Analysis of sample by one-dimensional electrophoresis is the most common form of protein gel electrophoresis.
- For separation and analysis of hundreds to thousands of proteins in one gel, a powerful electrophoretic method called two-dimensional gel electrophoresis is used.
- 2D gel electrophoresis separates a mixture of proteins according to two properties, one in each dimension.
- The first dimensions involve the separation based on native isoelectric point (pI), using form of electrophoresis called isoelectric focusing (IEF).
- Second dimensions separate mass using SDS-PAGE.
- This technique provides highest resolution for the protein analysis.
Reference and sources
- https://www.biotechnologynotes.com/electrophoresis/electrophoresis-meaning-definition-and-classification-withdiagram/293
- https://www.researchgate.net/publication/310994699_Fundamentals_and_Techniques_of_Biophysics_and_Molecular_Biology
- http://people.umass.edu/~mcclemen/581Proteins.html
- https://www.researchgate.net/publication/224829868_Gel-Electrophoresis_and_Its_Applications
- https://www.thermofisher.com/my/en/home/life-science/protein-biology/protein-biology-learning-center/proteinbiology-resource-library/pierce-protein-methods/overview-electrophoresis.html
- https://microbenotes.com/polyacrylamide-gel-electrophoresis-page/
- https://windinthewillowstudio.com/middlesex-centre/applications-of-polyacrylamide-gel-electrophoresis.php
- https://cdn.intechopen.com/pdfs/35089/InTech-Principles_of_nucleic_acid_separation_by_agarose_gel_electrophoresis.pdf
Also Read
- Southern Blotting
- Bacteria: Shape, Size, Structure and other Membrane
- Whole-Genome Shotgun Sequencing: overview, steps and achievements
- Vector: properties, types and characteristics
- Chromatography: Introduction, Principle, Classification and applications
- Different Types of chromatography
- SDS-PAGE: Introduction, Principle, Working and Steps
- Privacy Policy
- Application of Microbiology in Daily Life: Examples, Uses & Importance
- Microbial Identification and Strain Typing Using Molecular Techniques



4 thoughts on “Gel electrophoresis: types, principles, instrumentation and applications”
Well crafted… Very helpful
very nice written about this topic very easy to understand and helpful.
This blog post on gel electrophoresis is incredibly informative! I appreciate the detailed explanation of the different types and their specific applications in microbiology. The insights into the instrumentation used are also very helpful for someone looking to understand how these processes work in a lab setting. Thanks for breaking down such a complex topic!
This blog post on gel electrophoresis is incredibly informative! I appreciate the clear explanations of the different types and their applications in microbiology. The details on instrumentation were particularly helpful for understanding the setup. Great job breaking down such a complex topic!