Introduction
Penicillin is one of the earliest discovered and widely used antibiotics. It belongs to the β-lactam group of antibiotics and is derived from the mold Penicillium notatum (and P. chrysogenum). Discovered by Alexander Fleming in 1928, it marked the beginning of the antibiotic era.
- 1st antibiotic to be produced and still one of the best antibiotic available.
- Penicillin is a group of antibiotics, isolated from common molds known as Penicillium Molds.
- It is active against many gram-positive bacteria, Nocardia, Actinomycetes.
- It generally, interferes with cell wall synthesis of organisms which is sensitive and it is active only against vegetative cell.
History of Penicillin
- Sir, Alexander Fleming in 1929, first observed the antibiotic properties and possible therapeutics value of penicillin.
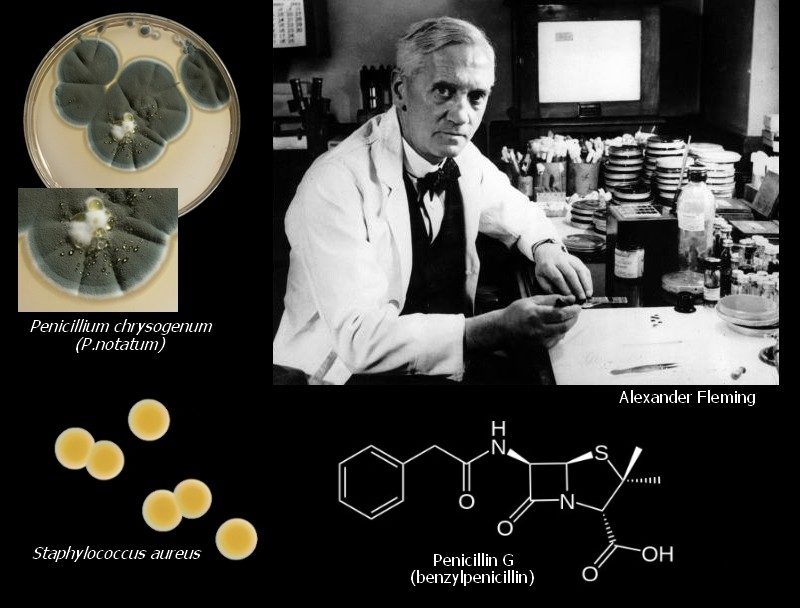
- He observed that an airborne contaminant, later shown to be Penicillium notatum inhibited the growth of a culture of Staphylococcus aureus on agar medium in the area surrounding the fungal activity.
- Fleming further showed that this inhibitory effect Caused by a soluble, diffusible mold product.
- Penicillin observed by Fleming was Penicillin-G.
Structure of Penicillin
- Several penicillins are closely related in structure.
- Common chemical nucleus and a side chain attached to this nucleus.

- Various compounds resembling the side chain can be added as precursor to fermentation medium & through microbial strain are directly incorporated into Penicillin molecule.
- Fungi: Aspergillus & Penicillium.
- Various penicillin differs as nature of their “R’’ side chain.
Penicillin molecule and its Precursor
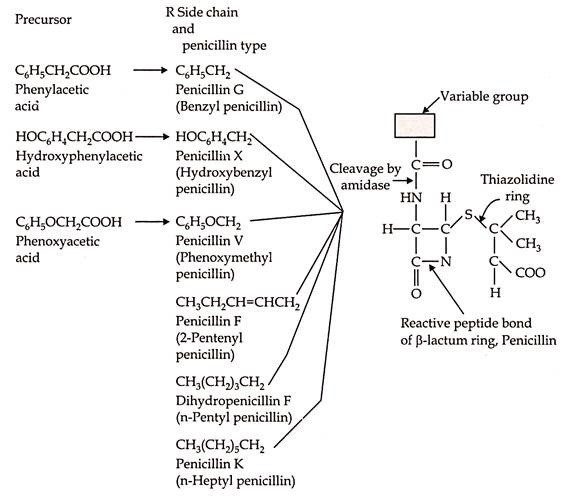
- Whereas, in present day commercial terms penicillin is regarded as the Penicillin-G.
- The Penicillin nucleus consists of two amino acids, L-cysteine and D-valine.
Penicillinase
- Penicillinase is an extracellular enzyme adaptively produced by members of the coliform group of bacteria, by most Bacillus sp.
- Major factor for penicillin resistance during infection.
- This enzyme hydrolyses penicillin to penicilloic acid.
6-amino penicillanic acid
- Sensitive to Penicillinase enzyme.
- Can be recovered from fermentation broth.
- Chemically converted to penicillin by chemical addition of various R side chains. Yield of this material is low.
- Penicillin acylase enzyme cleaves side chains of penicillin so resulted in commercial production 6-Amino penicillanic acid.
Penicillin production
- Commercially, penicillin was first produced in stationary mat culture.
- Production of penicillin is previously achieved by surface process i.e. solid-state fermentations and surface liquid fermentation.

Penicillin production mainly consists of three steps:
-
- Inoculum preparation
- Production fermentation
- Product recovery
Inoculum preparation
- Inoculum: Penicillium chrysogenum.
- For inoculum production, spores from heavily sporulated working stocks (special agar sporulation) are suspended in water or in a dilute solution of a nontoxic wetting agent, such as 1:10,000 sodium lauryl sulfonate.
- These spores are then poured in the flask which contains wheat bran and nutrient solution for heavy sporulation.
- Incubate for at 24°C for 5-7 days to provide heavy sporulation.
- Resulting spores are directly inoculated into inoculation tanks.
- Incubated at 25 to 26°C for 3 to 5 days exact duration of fermentation depends on various factors.
Production fermentation
- Method: fed batch or batch.
- Media: (Jackson 1958)
- Corn steep liquor solids-3.5%
- Lactose: 3.5%
- Cellulose: 1%
- CaCO3: 1%
- K2HPO4: 0.4%
- Edible oil: 0.25%
- Fermenter: stirred tank or air lift tank.
- Temperature: 25-27°C.
- Aeration: 0.5 – 27 1 VVM.
- Anti-foam: edible oil
- pH: set at 5.5 to 6.0 which increased up to 7-7.5.
- Inoculum media is same as production media.
- Lactose precursor is not added, at start of fermentation.
- PH remain constant, glucose, corn steep liquor, Carbon compounds and ammonia are utilised.
- Carbon compounds depleted, pH rise to 7-7.5.
- At the end of fermentation pH rises to 8 higher because depletion of lactose brings an autolysis in mycelium.
- Penicillium is harvested before this.
Product Recovery
- Harvest broth from fermenter tank by filtration.
- Chill to 5-10°C.
- Acidify filtrate to pH 2.0 to 2.5 with H2SO4.
- Penicillin extracted from aqueous filtrate into butyl acetate or amyl acetate.

- Discard aqueous fraction
- Add potassium acetate to the solvent extract in a crystallization tank to crystallize as potassium salt.
- Recover crystal from centrifuge.
- Further processing.
- Packaging.
Reference and Sources
- 1% – https://www.slideshare.net/guest08f61f/project-antibioticsbyharkiran-presentation
- 1% – https://quizlet.com/182638247/microbiology-iclicker-smartwork5-quiz-1-flash-cards/
- 1% – https://www.researchgate.net/publication/11010850_Optimization_of_a_fermentation_m
edium_for_the_production_of_Penicillin_G_acylase_from_Bacillus_sp - 1% – https://mafiadoc.com/download/practical-fermentation-technology_59da452a1723dd5e
e2b54f83.html
Also read:
- Ethanol : production, recovery and uses
- Microbiology Disciplines: Bacteria, Viruses, Fungi, Archaea and Protists
- Antimicrobial Chemotherapy: History, Characteristics and Tests
- Citric acid: Introduction, Fermentation, Recovery and Uses
- Streptomycin : chemical structure, production , recovery and uses.


1 thought on “Penicillin: History, Structure, Production and Recovery”
Kindly send the brief notes for penicillin production