Introduction
A Microbial Fuel Cell (MFC) is a bio-electrochemical system that uses microorganisms to convert chemical energy stored in organic compounds (like wastewater or biomass) directly into electrical energy. It offers a sustainable way to treat waste and generate green power simultaneously. A Microbial Fuel Cell (MFC) is a device that uses electrochemically active bacteria to convert organic matter into electricity through oxidation-reduction reactions.
Basic Principle
Microorganisms oxidize organic substrates (like glucose, acetate, or wastewater).
This releases electrons and protons:
- Electrons flow through an external circuit → generate electricity
- Protons (H⁺) pass through a proton exchange membrane (PEM) to the cathode
- At the cathode, electrons + protons + oxygen → water
Examples of Electroactive Bacteria
- Shewanella oneidensis
- Geobacter sulfurreducens
- Pseudomonas aeruginosa
MFCs Assembly
- An exciting and emerging field in microbiology is the use of bacteria to generate electricity, not through the production of methane but by directly capturing electrons from the microbe’s electron transport chain (ETC).
- This is possible because when some heterotrophic microorganisms oxidize organic material, they pass the electrons directly to an electrode a microbial fuel cell (MFC) captures these electrons to generate electricity.
- To accomplish this, the microbes are continuously fed a rich diet of organic substrates so that very little biosynthesis is required (recall that anabolic reactions require reducing power).
- In this way, most of the organic substrate is oxidized during catabolism and electrons are donated to the ETC.
- The basic design of a MFC is very simple.

- It consists two chambers, one oxic and the other anoxic, divided by a membrane through which protons can pass.
- Biomass is delivered to the bacteria in the anoxic side.
- The oxidation of this organic matter yields protons and electrons. The protons diffuse across the membrane to the oxic chamber. The electrons are deposited on an anode that connects the two chambers.
- Electrons flow toward the cathode but can be used to generate electricity while in transit. A catalyst in the oxic chamber combines the protons, electrons, and oxygen to generate water.
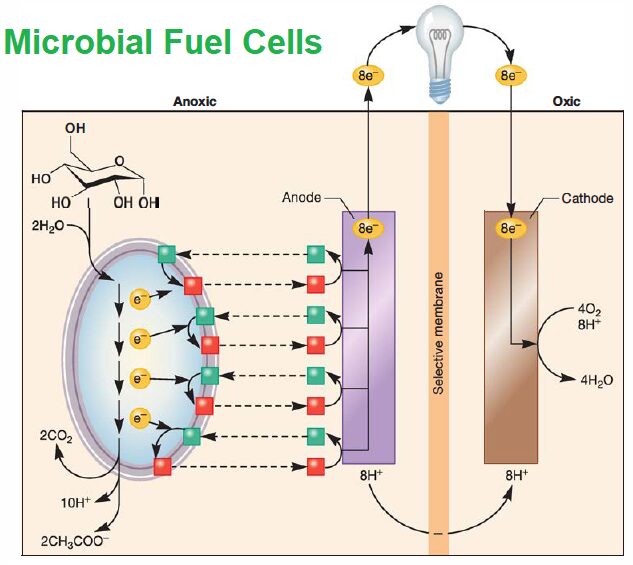
- Many different types of heterotrophic microbes can produce power in MFCs. The only requirement is that they oxidize organic matter anaerobically.
- In fact, communities of microbes can be employed. The largest constraint to obtaining maximum efficiency appears to be delivery of electrons to the electrode.
- There are several ways this can be accomplished. Often a chemical mediator is used to shuttle electrons from inside the cell, across the cellular membrane, to the anode.
- Some microbes, such as the γ-proteobacterium Shewanella oneidensis, produce “nano-wires” to transmit electrons to the anode (in nature, these nano-wires shuttle electrons to external electron acceptors such as Fe3+.
- MFC development is ongoing, and great strides have been made to maximize power output, which has increased orders of magnitude in just a decade.
Applications of microbial Fuel Cells
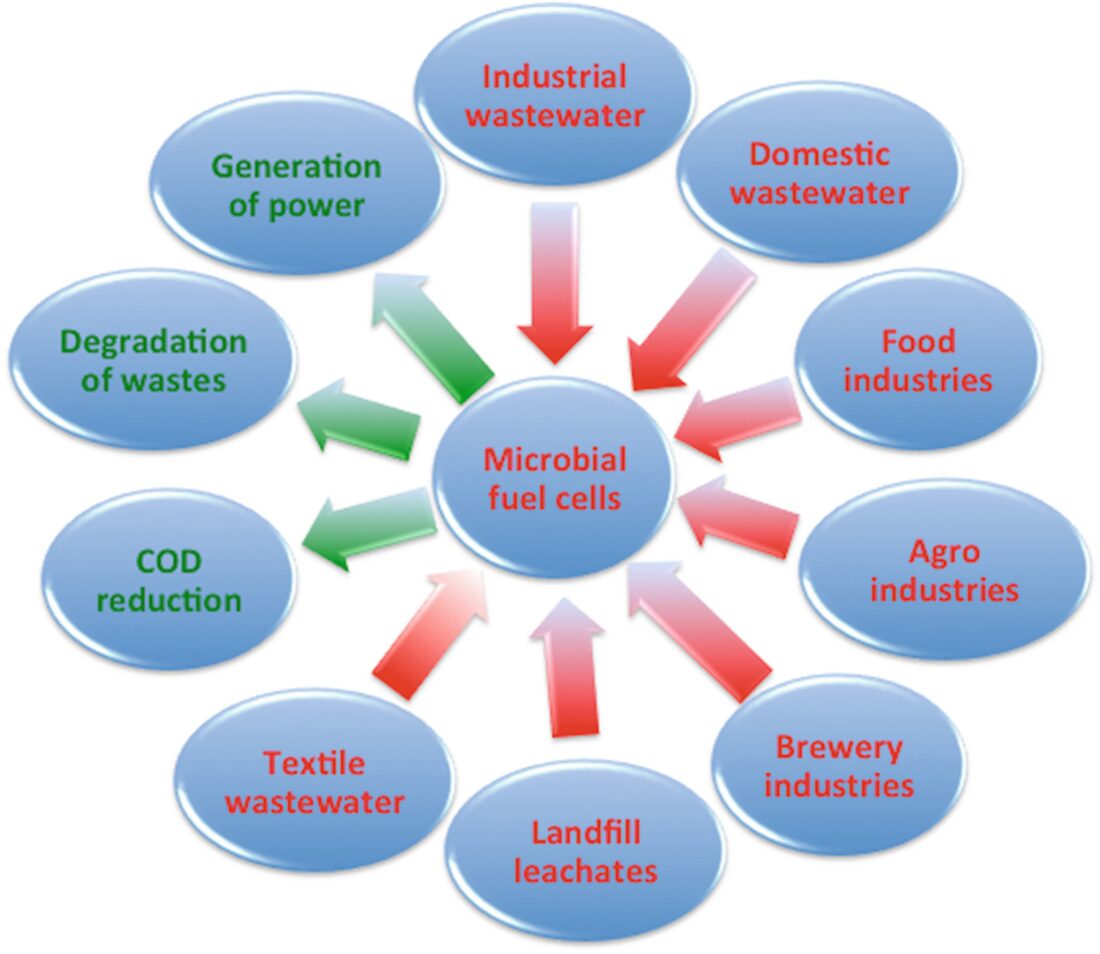
- MFCs include drinking water purification and the treatment of landfill leachate: in both cases, unwanted organic material is used to feed the bacteria in the MFC.
- MFCs can also be used when energy requirements are small but batteries cannot be used.
- Environmental monitoring instruments that need to be left in the field for years at a time are good examples.
- In addition, because MFCs are self-contained, they are attractive for developing countries that lack well-developed power grids.
Reference and sources
- https://www.researchgate.net/publication/294258512_Microbial_extracellular_electron_transfer_and_its_relevance_to_iron_corrosion
- https://www.researchgate.net/publication/233956244_Sulfide-Driven_Microbial_Electrosynthesis
- https://proteomics-conference.blogspot.com/2018/05/

