Introduction
- Hypothetically, the first living organism came into existence, when the atmosphere was devoid of oxygen, to drive the cell metabolism; anaerobic degradation of glucose was carried out by the process called Fermentation.
- When we talk about glucose catabolism, its pathways differ in two different conditions.
In aerobic state:
- Pyruvate is derived by glycolysis, later oxidized into acetate and enters the Kreb’s Cycle, oxidation of acetate forms water & carbon dioxide, and NADH which is reoxidized into NAD [electron donor] by oxygen, which enters the electron transport chains.
In anaerobic state:
- Unlike in aerobic respiration, NADH oxidation is not initiated due to devoid of oxygen, which creates a condition in which cells lack the electron acceptor, eventually halts the glycolysis pathway, to generate energy in such conditions, cells have adapted the strategy to regenerate NAD, through anaerobic glycolysis also termed as Fermentation.
- During the fermentation, NADH transfers electrons, to make reduced end products, like lactate or ethanol.
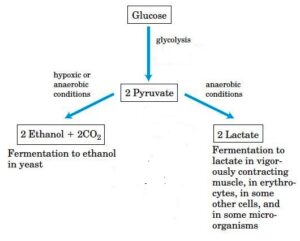
Types of the Fermentation process used by cells
Lactic Acid Fermentation
- When a sufficient amount of oxygen is not furnished in the animal tissues, NAD is regenerated, with a production of lactate, a reduced form of pyruvate.
- Cells such as erythrocytes [in which mitochondria are absent], which can’t oxidize pyruvate to CO2, forms lactate even under aerobic conditions.

- When 2 molecules of pyruvate are reduced, form 2 molecules of lactate regenerates 2 molecules of NAD, from the equation, we can depict there is no net change in NADH or NAD, and here electron acceptor is pyruvate.
- During vigorous muscular activity or exercise, skeleton muscles form lactate, which is recycled, bypassed along with the blood to the liver; during recovery of muscles, it is converted into glucose.
- Severe muscular contraction is experienced by an individual during a sprint or any other strenuous muscular activity due to the formation of a large amount of lactate.
- The above-discussed process is termed lactic acid fermentation, from which energy is obtained in the form of ATP, oxygen is not involved and no change in concentrations of NADH or NAD.
- The broad range of organisms that generally occupies anaerobic habitat carries out a fermentation process for their energy production; these organisms are commercially exploited to yield various end products.
Ethanol Fermentation
- Rather than opting lactic acid fermentation process, some microorganisms and yeasts ferment glucose into ethanol and carbon dioxide.
- By the glycolytic process, pyruvate is formed, which is later converted into ethanol and carbon dioxide by the process of ethanol fermentation.
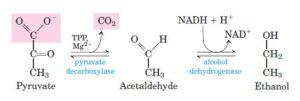
In the above reaction, pyruvate is transformed to ethanol by a two-step process:
- First, enzyme pyruvate decarboxylase, decarboxylate the pyruvate i.e., removal of the CO2 and form acetaldehyde that is carried out by the process of decarboxylation without any involvement of the oxidation. Mg2+ and coenzyme TPP [Thiamine Pyrophosphate] help the enzyme to carry out the process.
- In the second step, ethanol, a reduced form of acetaldehyde is formed by the enzyme alcohol dehydrogenase, with the reducing power of NADH.
- The end product of ethanol fermentation is ethanol and carbon dioxide.
Overall Reaction of Ethanol Fermentation
Glucose + 2ADP + 2Pi ——–> 2 Ethanol+ 2CO2+ 2ATP+ 2H2O
- Microorganisms and yeasts can carry ethanol fermentation as their source of energy, and essential enzymes such as pyruvate decarboxylase are present in their cells. Therefore on general basis yeasts are used in baking, for the fermentation of the dough. One more important enzyme is alcohol dehydrogenase, which is present in most organisms which include humans, action of the enzyme is carried in the liver when ethanol is either ingested or produced by microbiota present in the intestines.
References and Other Sources
- https://edurev.in/course/quiz/attempt/-1_Test-Respiration-Enzymes-1/918a90e4-490d-472fac89-3c3899e285b9
- https://www.ncbi.nlm.nih.gov/books/NBK22593
- https://laboratoryinfo.com/glycolysis-steps-diagram-energy-yield-and-significance
Also Read:
- Beer brewing process
- Different types of Pathways for ATP Production
- Chlamydia: Introduction, Epidemiology, Transmission, Treatment and Prevention
- Membrane Transport: Principle, Passive & Active Transport and Types
- Cider: Production, Extraction, Fermentation and Maturation
- Influence of Environmental Factors on Microbial Growth

