Vaccines: Definition, Types & Functions
Definition
Vaccines are substances that prepare the immune system to recognize and respond to a pathogen (antigen), resulting in protection (immunity) to that pathogen. Vaccines are simply “educating” the body’s natural infection fighting system. They are composed of treated microorganisms or viruses, chemically altered toxins, or molecular parts of microorganisms, and the vaccines usually are administered by injection or oral consumption.
Most vaccines represent preventative vaccines, that is, vaccines that protect an individual from contracting a particular infectious disease. A few vaccines represent therapeutic vaccines, which act to lessen the severity of the illness that might appear or already exists.
For example, shingles can be a very painful and debilitating disease. Getting the shingles vaccine will not protect one from getting shingles (the viral DNA already is present in the body), but the vaccine will lessen the severity and length of symptoms should shingles occur.
Although we will use the term “vaccination” and “immunization” are not quite the same thing. Vaccination is the process of administering a vaccine that produces immunity in the body. Immunization is the process by which an individual becomes protected from an infectious disease.
Different types of vaccines
Whole-Agent Vaccines Weakened or Inactivated Antigens
Attenuated Vaccines
Some viral and bacterial pathogens can be weakened in the pharmaceutical lab such that they should not cause disease. These attenuated pathogens multiply only at low rates in the body, increasing the dose of antigen to a level that will stimulate an immune response. Such vaccines are the closest to the natural pathogens and, because they most closely mimic a natural infection, they generate the strongest immune response. Often, the person vaccinated will have lifelong immunity.
The downside of attenuated vaccines results from the slow but continued multiplication of the bacterium or virus composing the vaccine. Because the vaccines contain dividing bacterial cells or replicating viruses, there is a remote chance one of them could increase in population size to cause a mild form of the disease. Usually a healthy person
with a fully functioning immune system (immuno competent) clears the infection without serious consequence.
However, infants, older adults, and individuals with a compromised immune system, such as patients with AIDS or pregnant women, should not be given attenuated vaccines, if possible. Today, many vaccines, such as the Sabin oral polio vaccine and the measles, mumps, and chickenpox vaccines, contain attenuated viruses.
To avoid multiple injections of immunizing agents, it sometimes is advantageous to combine vaccines into a single-dose vaccine. The measles mumps-rubella (MMR) vaccine is one example. Attenuated vaccines can be destroyed by heat, so they must be refrigerated to retain their effectiveness. Consequently, on a global scale, attenuated vaccines might not be the vaccine strategy of choice, because many developing nations lack widespread refrigeration facilities.
Inactivated Vaccines
Another strategy for preparing vaccines is to “kill” the bacterial or viral pathogen. These inactivated vaccines are relatively easy to produce because the pathogen is killed by simply using specific chemicals and/or heat. Although the agents in these vaccines cannot reproduce or replicate, the inactivation process alters the antigen’s structure and shape. This results in a weaker humoral immune response and little or no cell-mediated immunity.
For this reason, most inactivated vaccines require multiple doses to generate a protective response, and periodic supplemental doses, or booster shots, might be needed throughout life to maintain immunity. The Salk polio vaccine and the hepatitis A vaccine typify such preparations of inactivated whole viruses. In the case of influenza, the virus changes genetically from year to year, so a different vaccine must be provided annually.
Compared to attenuated vaccines, inactivated vaccines are safer because they cannot cause the disease in a vaccinated individual. The vaccines can be stored in a freeze-dried form at room temperature, making them a vaccine of choice in developing nations.
Toxoid Vaccines
For some bacterial diseases, such as diphtheria and tetanus, a bacterial exotoxin is the main cause of illness. For these diseases, a third immunization strategy is to inactivate these toxins and use them as a vaccine. Such toxins can be inactivated with formalin (an aqueous solution of formaldehyde) and the resulting inactivated toxin is called a toxoid.
Immunity induced by a toxoid vaccine allows the body to generate antibodies and memory cells to recognize
the natural toxin, should the individual be exposed to the active toxin. Because toxoid vaccines are inactivated products, booster shots are necessary.
Single-dose vaccines include diphtheria pertussis tetanus (DPT) vaccine and the newer diphtheria-tetanus-acellular pertussis (DTaP) vaccine. For other vaccines, however, a combination single-dose vaccine might not be useful because the antibody response might be lower for the combination than for each vaccine taken separately.
Newer Vaccines Contain Only Subunits or Fragments of Antigens
So-called second-generation vaccines have been developed that contain only a subunit or fragment of the bacterial cell or virus. With only a small piece of the antigen, a vaccinated individual cannot contract the disease from the vaccine, but these vaccines might fail to generate a cytotoxic T-cell response as part of adaptive immunity.
Subunit Vaccines
Unlike the whole-agent vaccines, the strategy for a subunit vaccine is to have the vaccine contain only those parts or subunits of the antigen that stimulate a strong immune response. For example, the subunit vaccine for pneumococcal pneumonia contains 23 different polysaccharides from the capsules of 23 strains of Streptococcus pneumoniae.
One way of producing a subunit vaccine is to use recombinant DNA technology, in which the resulting vaccine is called a recombinant subunit vaccine. The hepatitis B vaccine (Recombivax HB® and Engerix-B®) are examples. Several hepatitis B virus genes are isolated and inserted into yeast cells, which then synthesize the hepatitis antigens. These antigens are collected and purified to make the vaccine. Adverse reactions to such subunit vaccines are very rare, and the subunits cannot produce disease in the person vaccinated.
Conjugate Vaccines
Haemophilus influenzae b (Hib), which is responsible for a form of childhood meningitis, produces a capsule. Materials, like the capsular polysaccharides, represent haptens, which refers to substances that are not strongly immunogenic; that is, they do not provoke a strong immune response.
Therefore, the strategy is to conjugate (attach) the haptens to a protein molecule, such as the tetanus or diphtheria toxoid that will stimulate a strong immune response. The result is the Hib conjugate vaccine, which has been instrumental in reducing the incidence of Haemophilus meningitis in young American children (younger than 5 years of age) from 18,000 cases annually in 1986 to 22 cases in 2015.
Presents the 2016 immunization schedule for children 0 to 6 years of age as recommended by the Centers for Disease Control and Prevention (CDC).
DNA Vaccines
Part of the renaissance in vaccine technology is a strategy to use DNA as a vaccine. These investigational DNA vaccines consist of plasmids engineered to contain one or more protein-encoding genes from a viral or bacterial pathogen. By being injected intramuscularly in a saline solution, cells in the body will take up the injected foreign
DNA, express (transcribe and translate) the proteins encoded by the DNA, and display these antigens on the cell surface, much like an infected cell presents antigen peptides to T cells.
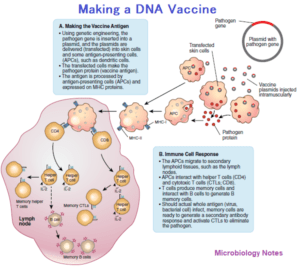
Such a display should stimulate a strong antibody (humoral) and cell-mediated immune response. If the vaccinated individual encounters the actual pathogen later, the individual should be immune to contracting the disease.
Thus far, few experimental trials have produced an immune response equivalent to the other forms of vaccines. However, in July 2005, a veterinary DNA vaccine to protect horses from West Nile virus became the world’s first licensed DNA vaccine. In terms of humans, plasmid-based DNA vaccines are being studied for a variety of infections, including AIDS, influenza, hepatitis C, and for several cancers.
Some Vaccines Recommended for Adults
Many adults are unaware (or forget) that they need vaccines or booster shots throughout their lives. As a result, adult vaccination rates in the United States are unacceptably low. In fact, more than 40,000 adults in the United States die each year from diseases that are vaccine preventable. Presents the CDC’s immunization schedule for adults (19 years and older).
There are several reasons why adult vaccinations are needed. First, some adults might not have experienced a childhood disease such as chickenpox or measles, or perhaps they were never vaccinated as children. Adult chickenpox can lead to serious pneumonia complications, whereas adult measles can produce serious complications, including arthritis and encephalitis (brain inflammation).
Second, newer vaccines, such as the hepatitis B and shingles vaccines, might not have been available when some adults were younger. Shingles is a rare illness in children, so the CDC recommends that adults 60 years and older get the shingles vaccine.
Third, as adults get older and their immune systems begin to decline in strength, they become more susceptible to serious diseases caused by common infections such as flu and tetanus. Fourth, some vaccines simply do not afford protection beyond one “infectious season.” The flu virus changes every season, so a vaccine taken one season might not protect for the next flu season.
One other reason for adult vaccinations stems from the fact that many vaccines do not provide lifelong immunity, and the immune system’s memory begins to fade after several years. Therefore, booster shots provide additional doses of a vaccine given periodically to “refresh” the immune system’s antibodies and memory cells.
Consequently, some form of either the tetanus-diphtheria (Td) vaccine or the tetanus-diphtheria-acellular pertussis (Tdap) vaccine is recommended for adults every 10 years. In some cases, killed pathogens do not stimulate a strong immune response, and substances need to be added to increase their effectiveness.
Reference and Sources
- https://www.whattoserveagoddess.com/tag/vaccines/
- https://immunize.org/askexperts/administering-vaccines.asp
- https://quizlet.com/348991186/immunization-and-serology-chapter-23-flash-cards/
- https://www.ninds.nih.gov/Disorders/Patient-Caregiver-Education/Fact-Sheets/Guillain-Barr%C3%A9-Syndrome-Fact-Sheet
- https://www.immunology.org/publications/bsi-reports/60-years-immunology-past-present-and-future/vaccinationprevention
- https://www.cdc.gov/vaccines/hcp/conversations/understanding-vacc-work.html
- https://quizlet.com/31038489/bio-205-exam-5-flash-cards/
- https://quizlet.com/107595053/micro-ch-22-immunity-and-serology-flash-cards/
- https://openstax.org/books/microbiology/pages/18-5-vaccines
- https://bio.libretexts.org/Bookshelves/Introductory_and_General_Biology/Book%3A_Biology_(Kimball)/15%3A_The_Anatomy_and_Physiology_of_Animals/15.04%3A_Immune_System/15.4W%3A_Vaccines
- https://quizlet.com/163026304/micro-bio-ch-17-immunity-and-serology-flash-cards/
- https://www.cdc.gov/media/pressrel/r050718.html
- https://www.ncbi.nlm.nih.gov/books/NBK26926/
- https://www.who.int/news-room/feature-stories/detail/how-do-vaccines-work
